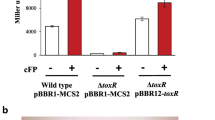
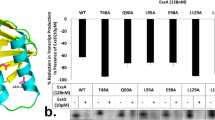
Abstract
The Shigella flexneri outer membrane protease IcsP proteolytically cleaves the actin-based motility protein IcsA from the bacterial surface. The icsP gene is monocistronic and lies downstream of an unusually large intergenic region on the Shigella virulence plasmid. In silico analysis of this region predicts a second transcription start site 84 bp upstream of the first. Primer extension analyses and beta-galactosidase assays demonstrate that both transcription start sites are used. Both promoters are regulated by the Shigella virulence gene regulator VirB and both respond similarly to conditions known to influence Shigella virulence gene expression (iron concentration, pH, osmotic pressure, and phase of growth). The newly identified promoter lies upstream of a Shine–Dalgarno sequence and second 5′-ATG-3′, which is in frame with the annotated icsP gene. The use of either translation start site leads to the production of IcsP capable of proteolytically cleaving IcsA. A bioinformatic scan of the Shigella genome reveals multiple occurrences of in-frame translation start sites associated with putative Shine–Dalgarno sequences, immediately upstream and downstream of annotated open reading frames. Taken together, our observations support the possibility that the use of in-frame translation start sites may generate different protein isoforms, thereby expanding the proteome encoded by bacterial genomes.






Similar content being viewed by others
References
Aiba H, Adhya S, de Crombrugghe B (1981) Evidence for two functional gal promoters in intact Escherichia coli cells. J Biol Chem 256:11905–11910
Akita M, Sasaki S, Matsuyama S, Mizushima S (1990) SecA interacts with secretory proteins by recognizing the positive charge at the amino terminus of the signal peptide in Escherichia coli. J Biol Chem 265:8164–8169
Ambion (2001) DNase I Demystified. TechNotes Newsletter 8
Bernardini ML, Mounier J, d’Hauteville H, Coquis-Rondon M, Sansonetti PJ (1989) Identification of icsA, a plasmid locus of Shigella flexneri that governs bacterial intra- and intercellular spread through interaction with F-actin. Proc Natl Acad Sci USA 86:3867–3871
Blattner FR, Plunkett G III, Bloch CA, Perna NT, Burland V, Riley M, Collado-Vides J, Glasner JD, Rode CK, Mayhew GF, Gregor J, Davis NW, Kirkpatrick HA, Goeden MA, Rose DJ, Mau B, Shao Y (1997) The complete genome sequence of Escherichia coli K-12. Science 277:1453–1462
Brandt F, Etchells SA, Ortiz JO, Elcock AH, Hartl FU, Baumeister W (2009) The native 3D organization of bacterial polysomes. Cell 136:261–271
Castellanos MI, Harrison DJ, Smith JM, Labahn SK, Levy KM, Wing HJ (2009) VirB alleviates H-NS repression of the icsP promoter in Shigella flexneri from sites more than one kilobase upstream of the transcription start site. J Bacteriol 191:4047–4050
d’Hauteville H, Dufourcq Lagelouse R, Nato F, Sansonetti PJ (1996) Lack of cleavage of IcsA in Shigella flexneri causes aberrant movement and allows demonstration of a cross-reactive eukaryotic protein. Infect Immun 64:511–517
Delcher AL, Harmon D, Kasif S, White O, Salzberg SL (1999) Improved microbial gene identification with GLIMMER. Nucleic Acids Res 27:4636–4641
Dorman CJ, Porter ME (1998) The Shigella virulence gene regulatory cascade: a paradigm of bacterial gene control mechanisms. Mol Microbiol 29:677–684
Egile C, d’Hauteville H, Parsot C, Sansonetti PJ (1997) SopA, the outer membrane protease responsible for polar localization of IcsA in Shigella flexneri. Mol Microbiol 23:1063–1073
Emanuelsson O, Brunak S, von Heijne G, Nielsen H (2007) Locating proteins in the cell using TargetP, SignalP and related tools. Nat Protoc 2:953–971
Erickson JW, Vaughn V, Walter WA, Neidhardt FC, Gross CA (1987) Regulation of the promoters and transcripts of rpoH, the Escherichia coli heat shock regulatory gene. Genes Dev 1:419–432
Fekkes P, Driessen AJ (1999) Protein targeting to the bacterial cytoplasmic membrane. Microbiol Mol Biol Rev 63:161–173
Gaal T, Ross W, Estrem ST, Nguyen LH, Burgess RR, Gourse RL (2001) Promoter recognition and discrimination by EσS RNA polymerase. Mol Microbiol 42:939–954
Goldberg MB, Barzu O, Parsot C, Sansonetti PJ (1993) Unipolar localization and ATPase activity of IcsA, a Shigella flexneri protein involved in intracellular movement. Infect Agents Dis 2:210–211
Goldberg MB, Theriot JA, Sansonetti PJ (1994) Regulation of surface presentation of IcsA, a Shigella protein essential to intracellular movement and spread, is growth phase dependent. Infect Immun 62:5664–5668
Jin Q, Yuan Z, Xu J, Wang Y, Shen Y, Lu W, Wang J, Liu H, Yang J, Yang F, Zhang X, Zhang J, Yang G, Wu H, Qu D, Dong J, Sun L, Xue Y, Zhao A, Gao Y, Zhu J, Kan B, Ding K, Chen S, Cheng H, Yao Z, He B, Chen R, Ma D, Qiang B, Wen Y, Hou Y, Yu J (2002) Genome sequence of Shigella flexneri 2a: insights into pathogenicity through comparison with genomes of Escherichia coli K12 and O157. Nucleic Acids Res 30:4432–4441
Labrec EH, Schneider H, Magnani TJ, Formal SB (1964) Epithelial cell penetration as an essential step in the pathogenesis of bacillary dysentery. J Bacteriol 88:1503–1518
Le Gall T, Mavris M, Martino MC, Bernardini ML, Denamur E, Parsot C (2005) Analysis of virulence plasmid gene expression defines three classes of effectors in the type III secretion system of Shigella flexneri. Microbiology 151:951–962
Makino S, Sasakawa C, Kamata K, Kurata T, Yoshikawa M (1986) A genetic determinant required for continuous reinfection of adjacent cells on large plasmid in S. flexneri 2a. Cell 46:551–555
Miller J (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory Press, New York
Mitobe J, Morita-Ishihara T, Ishihama A, Watanabe H (2009) Involvement of RNA-binding protein Hfq in the osmotic-response regulation of invE gene expression in Shigella sonnei. BMC Microbiol 9:110
Murphy ER, Payne SM (2007) RyhB, an iron-responsive small RNA molecule, regulates Shigella dysenteriae virulence. Infect Immun 75:3470–3477
Porter ME, Dorman CJ (1994) A role for H-NS in the thermo-osmotic regulation of virulence gene expression in Shigella flexneri. J Bacteriol 176:4187–4191
Porter ME, Dorman CJ (1997) Differential regulation of the plasmid-encoded genes in the Shigella flexneri virulence regulon. Mol Gen Genet 256:93–103
Pupo GM, Lan R, Reeves PR (2000) Multiple independent origins of Shigella clones of Escherichia coli and convergent evolution of many of their characteristics. Proc Natl Acad Sci USA 97:10567–10572
Raina S, Missiakas D, Georgopoulos C (1995) The rpoE gene encoding the σE (σ24) heat shock sigma factor of Escherichia coli. EMBO J 14:1043–1055
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, New York
Sansonetti PJ (1998) Molecular and cellular mechanisms of invasion of the intestinal barrier by enteric pathogens. The paradigm of Shigella. Folia Microbiol (Praha) 43:239–246
Shere KD, Sallustio S, Manessis A, D’Aversa TG, Goldberg MB (1997) Disruption of IcsP, the major Shigella protease that cleaves IcsA, accelerates actin-based motility. Mol Microbiol 25:451–462
Steinhauer J, Agha R, Pham T, Varga AW, Goldberg MB (1999) The unipolar Shigella surface protein IcsA is targeted directly to the bacterial old pole: IcsP cleavage of IcsA occurs over the entire bacterial surface. Mol Microbiol 32:367–377
Tucker AC, Escalante-Semerena JC (2010) Biologically active isoforms of CobB Sirtuin deacetylase in Salmonella enterica and Erwinia amylovora. J Bacteriol 192:6200–6208
Vandeputte-Rutten L, Kramer RA, Kroon J, Dekker N, Egmond MR, Gros P (2001) Crystal structure of the outer membrane protease OmpT from Escherichia coli suggests a novel catalytic site. EMBO J 20:5033–5039
Wei C, Yang J, Zhu J, Zhang X, Leng W, Wang J, Xue Y, Sun L, Li W, Wang J, Jin Q (2006) Comprehensive proteomic analysis of Shigella flexneri 2a membrane proteins. J Proteome Res 5:1860–1865
Wing HJ, Williams SM, Busby SJ (1995) Spacing requirements for transcription activation by Escherichia coli FNR protein. J Bacteriol 177:6704–6710
Wing HJ, Yan AW, Goldman SR, Goldberg MB (2004) Regulation of IcsP, the outer membrane protease of the Shigella actin tail assembly protein IcsA, by virulence plasmid regulators VirF and VirB. J Bacteriol 186:699–705
Wing HJ, Goldman SR, Ally S, Goldberg MB (2005) Modulation of an outer membrane protease contributes to the virulence defect of Shigella flexneri strains carrying a mutation in the virK locus. Infect Immun 73:1217–1220
Acknowledgments
We thank N. Ward and C. Ross for their assistance with this project. This study was supported by the National Institutes of Health (NIH) grant P20 RR-016464 from the IDeA Networks of Biomedical Research Excellence Program of the National Center for Research Resources grant, by the NIH grant R15 AI090573-01 and by UNLV start-up funds to H.J.W. K.M.L. was a 2009 Recipient of a Barry Goldwater Scholarship and C.T.H. is currently supported in part by the Post-9/11 GI Bill.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by John Helmann.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
Position-specific scoring matrix for ribosome binding sites associated with the S. flexneri 2a strain 301 virulence plasmid pCP301 (NC_004851.1) and chromosome (NC_004337.1). Six nucleotide-long motif overrepresented in immediate upstream regions of annotated start codons identified by MEME (http://www.meme.sdsc.edu) and used to search for Shine–Dalgarno sequences upstream of predicted additional start codons. (PPT 142 kb)
Online Resource 2
Distribution of p-values for predicted ribosome binding sites with respect to both the annotated translation start sites (black) and the predicted additional translation start sites (gray). (PPT 122 kb)
Rights and permissions
About this article
Cite this article
Hensley, C.T., Kamneva, O.K., Levy, K.M. et al. Two promoters and two translation start sites control the expression of the Shigella flexneri outer membrane protease IcsP. Arch Microbiol 193, 263–274 (2011). https://doi.org/10.1007/s00203-010-0669-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-010-0669-2