Abstract
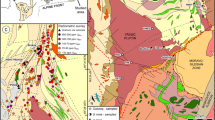
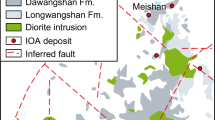
The Matongo carbonatite intrusive body in the Neoproterozoic Upper Ruvubu alkaline plutonic complex (URAPC) in Burundi is overlain by an economic phosphate ore deposit that is present as breccia lenses. The ore exhibits evidence of supergene enrichment but also preserves textures related to the concentration of fluorapatite in the carbonatitic system. Magmatic fluorapatite is abundant in the ore and commonly occurs as millimeter-sized aggregates. It is enriched in light rare earth elements (LREE), which is especially apparent in the final generation of magmatic fluorapatite (up to 1.32 wt% LREE2O3). After an episode of metasomatism (fenitization), which led to the formation of K-feldspar and albite, the fluorapatite-rich rocks were partly brecciated. Oxygen and carbon isotope compositions obtained on the calcite forming the breccia matrix (δ18O = 22.1 ‰ and δ13C = −1.5 ‰) are consistent with the involvement of a fluid resulting from the mixing of magmatic-derived fluids with a metamorphic fluid originating from the country rocks. In a subsequent postmagmatic event, the carbonates hosting fluorapatite were dissolved, leading to intense brecciation of the fluorapatite-rich rocks. Secondary carbonate-fluorapatite (less enriched in LREE with 0.07–0.24 wt% LREE2O3 but locally associated with monazite) and coeval siderite constitute the matrix of these breccias. Siderite has δ18O values between 25.4 and 27.7 ‰ and very low δ13C values (from −12.4 to −9.2 ‰), which are consistent with the contribution of organic-derived low δ13C carbon from groundwater. These signatures emphasize supergene alteration. Finally, the remaining voids were filled with a LREE-poor fibrous fluorapatite (0.01 wt% LREE2O3), forming hardened phosphorite, still under supergene conditions. Pyrochlore and vanadiferous magnetite are other minerals accumulated in the eluvial horizons. As a consequence of the supergene processes and fluorapatite accumulation, the phosphate ore, which contains 0.72 to 38.01 wt% P2O5, is also enriched in LREE (LaN/YbN from 47.1 to 83.5; ΣREE between 165 and 5486 ppm), Nb (up to 656 ppm), and V (up to 1232 ppm). In the case of phosphate exploitation at Matongo, REE could prove to have a subeconomic potential to be exploited as by-products of phosphates.







Similar content being viewed by others
References
Alves PR (2008) The carbonatite-hosted apatite deposit of Jacupiranga, SE Brazil: styles of mineralization, ore characterization, and association with mineral processing. Unpublished PhD thesis, Missouri University of Science and Technology
Andrade FRD, Möller P, Lüders V, Dulski P, Gilg HA (1999) Hydrothermal rare earth elements mineralization in the Barra do Itapirapuã carbonatite, southern Brazil: behaviour of selected trace elements and stable isotopes (C, O). Chem Geol 155:91–113
Ayers JC, Watson EB (1991) Solubility of apatite, monazite, zircon, and rutile in supercritical aqueous fluids with implications for subduction zone geochemistry. Philos Trans R Soc Lond Ser A Phys Eng Sci 335:365–375
Bambi AJCM, Costanzo A, Malgarejo JC, Conçalves AO, Alfonso P, Buta Neto A, Manuel J (2008) Evolution of pyrochlore in carbonatites: the Angola case. Revista de la sociedad española de mineralogía, pp 43–44
Barbarand J, Pagel M (2001) Cathodoluminescence study of apatite crystals. Am Mineral 86:473–484
Barros de Oliveira SM, Liguori Imbernon RA (1998) Weathering alteration and related REE concentration in the Catalão I carbonatite complex, central Brazil. J S Am Earth Sci 11:379–388
Bonev I (1972) On the terminology of the phenomena of mutual crystal orientation. Acta Crystallogr Sect A Cryst Phys, Diffr, Theor Gen Crystallogr 28:508–512
Boulvais P, Cobert C, Baele JM, Tack L, Nimpagaritse G, Gardien V, Demaiffe D, Decrée S (2015) A syntectonic alkaline massif in Burundi: geometry, fluid-rock interaction and element (REE, HFSE) mobility. 13th SGA biennal meeting, Nancy, France. Abstract book
Bouzari F, Hart C, Barker S, Bissig T (2014) Porphyry indicator minerals (PIMS): a new exploration tool for concealed deposits in south-central British Columbia. University of British Columbia. Geoscience BC Report 2011–17, 31 p
Carothers WW, Lanford HA, Rosenbauer RJ (1988) Experimental oxygen isotope fractionation between siderite-water and phosphoric acid liberated CO2-siderite. Geochim Cosmochim Acta 52:2445–2450
Chakhmouradian AR (2006) High-field-strength elements in carbonatitic rocks: geochemistry, crystal chemistry and significance for constraining the sources of carbonatites. Chem Geol 235:138–160
Davies KA (1956) The geology of part of south-east Uganda, with special reference to the alkaline complexes. Geol Surv Uganda Mem 8:62–76
De Putter T, Mees F, Ruffet G, Delvaux D, Decrée S, Dewaele S, Nimpagaritse G, Nkurunziza P, Tack L (2013) Exploring the Neogene as a period for supergene ore formation and exposure in the Great Lakes Region (Wester Great Rift Valley). 24th Colloquium of African Geology, Addis Ababa, Ethiopia. Abstract book: p 140
De Toledo MCM, Lenharo SL, Ferrari VC, Fontan F, De Parseval P, Leroy G (2004) The compositional evolution of apatite in the weathering profile of the Catalão I alkaline-carbonatitic complex, Goiás, Brazil. Can Mineral 42:1139–1158
Deans T (1966) Economic mineralogy of African carbonatites. In: Tuttle OF, Gittins J (eds) Carbonatites. Interscience Wiley, New York, pp 385–413
Decrée S, Deloule É, Ruffet G, Dewaele S, Mees F, Marignac C, Yans J, De Putter T (2010) Geodynamic and climate controls in the formation of Mio–Pliocene world-class oxidized cobalt and manganese ores in the Katanga province, DR Congo. Mineral Deposits 45(7):621–629
Decrée S, Boulvais P, Cobert C, Baele JM, Midende G, Gardien V, Tack L, Nimpagaritse G, Demaiffe D (2015) Structurally-controlled hydrothermal alteration in the Upper Ruvubu Alkaline Plutonic Complex (Burundi): implications for REE and HFSE mobilities. Precambrian Res 269:281–295
Delvaux D, Kervyn F, Macheyeki AS, Temu EB (2012) Geodynamic significance of the TRM segment in the East African Rift (W-Tanzania): active tectonics and paleostress in the Ufipa plateau and Rukwa basin. J Struct Geol 37:161–180
Demaiffe D (2008) Le magmatisme alcalin et carbonatitique: synthèse sur la province paléozoique de Kola (Russie) et caractéristiques générales du massif protérozoique de Matongo (Burundi). Bull Séances Acad R Sci O-M 54:171–196
Edou-Minko A (2003) Geochemical profile of the Mabounie niobiferous residual deposit (Gabon). J Sci 3:56–62
Emsbo P, McLaughlin PI, Breit GN, du Bray EA, Koenig AE (2015) Rare earth elements in sedimentary phosphate deposits: solution to the global REE crisis? Gondwana Res 27:776–785
Fagel N, André L, Debrabant P (1997) Multiple “seawater” derived geochemical signature in the Indian Ocean pelagic clays? Geochim Cosmochim Acta 61:989–1008
Fernandez-Alonso M, Cutten H, De Waele B, Tack L, Tahon A, Baudet D, Barritt SD (2012) The Mesoproterozoic Karagwe-Ankole Belt (formerly the NE Kibara Belt): the result of prolonged extensional intracratonic basin development punctuated by two short-lived far-field compressional events. Precambrian Res 216:63–86
Freyssinet P, Butt CRM, Morris RC, Piantone P (2005) Ore-forming processes related to lateritic weathering. Econ Geol 100th anniversary volume, pp 681–722
Ghorbani M (2013) The economic geology of Iran mineral deposits and natural resources. Springer Geology, Springer, 569 p
Gieré R (1990) Hydrothermal mobility of Ti, Zr and REE: examples from the Bergell and Adamello contact aureoles (Italy). Terra Nov. 2:60–67
Gieré R (1996) Formation of rare earth minerals in hydrothermal systems. In: Jones AP, Wall F, Williams CT (eds) Rare earth minerals, vol 7, The mineralogical series. Chapman & Hall, London, pp 105–150
Götte J (2009) Petrological modifications in continental target rocks from terrestrial impact structures: evidence from cathodoluminescence. In: Gucsik A (ed) Cathodoluminescence and its application in the planetary sciences, pp 45–60
Götze J, Kempe U (2009) Physical principles of cathodoluminescence and its applications to geosciences. In: Gucsik A (ed) Cathodoluminescence and its application in the planetary sciences. Springer, Heidelberg, pp 1–22
Habermann D, Neuser RD, Richter DK (2000) Quantitative high resolution spectral analysis of Mn2+ in sedimentary calcite. In: Pagel M, Barbin V, Blanc P, Ohnenstetter D (eds) Cathodoluminescence in geosciences. Springer, Heidelberg, pp 331–358
Harlov DE, Andersson UB, Förster HJ, Nyström JO, Dulski P, Broman C (2002) Apatite-monazite relations in the Kiirunavaara magnetite-apatite ore, northern Sweden. Chem Geol 191:47–72
Harlov DE, Wirth R, Förster HJ (2005) An experimental study of dissolution-reprecipitation in fluorapatite: fluid infiltration and the formation of monazite. Contrib Miner Petrol 150:268–286
Hayward CL, Jones AP (1991) Cathodoluminescence petrography of Middle Proterozoic extrusive carbonatite from Qasiarsuk, South Greenland. Min Mag 55:591–603
Kempe U, Götze J (2002) Cathodoluminecence (CL) behaviour and crystal chemistry of apatite from rare-metal deposits. Min Mag 66:151–172
Kurtanjek MP, Tandy BC (1989) The igneous phosphate deposits of Matongo-Bandaga, Burundi. In: Notholt AJG, Sheldon RP, Davidson DF (eds) Phosphate deposits of the world, vol 2, Phosphate rock resources. Cambridge University Press, Cambridge, pp 262–266
Lypaczewski P, Normandeau PX, Paquette J, McMartin I (2013) Petrographic and cathodoluminescence characterization of apatite from the Sue-Dianne and Brooke IOCG mineralization systems, Great Bear magmatic zone, Northwest Territories. Geol Surv Can, Open File 7319. doi:10.4095/292369
Mariano AM (1988) Some further geological application of cathodoluminescence. In: Marshall DJ (ed) Cathodoluminescence of geological materials. Unwin Hyman, Boston, pp 94–123
Mariano AN (1989) Nature of economic mineralization in carbonatites and related rocks. In: Bell K (ed) Carbonatites: genesis and evolution. Unwin Hyman, London, pp 149–176
Marshall DJ (1988) Cathodoluminescence of geological materials. Hyman, Boston, 146 p
McDonough WF, Sun SS, Ringwood AE, Jagoutz E, Hofmann AW (1992) Potassium, rubidium and cesium in the Earth and moon and the evolution of the mantle of the Earth. Geochim Cosmochim Acta 56:1001–1012
McLemore VT, Barker JM (1987) Some geological application of cathodoluminescence, examples from the Lemitar Mountains and Riley travertine, Socorro County, New Mexico. N M Geol, 37–40
Midende G (1984) La carbonatite de Matongo (Burundi). Unpublished PhD thesis, Université Libre de Bruxelles, Bruxelles
Midende G, Boulvais P, Tack L, Melcher F, Gerdes A, Dewaele S, Demaiffe D, Decrée D (2014) Petrography, geochemistry and U-Pb zircon age of the Matongo carbonatite Massif (Burundi): implication for the Neoproterozoic geodynamic evolution of Central Africa. J Afr Earth Sci 100:656–674
Mitchell RH (2014) Primary and secondary niobium mineral deposits associated with carbonatites. Ore Geol Rev 64:626–641
Nasraoui M, Bilal E (2000) Pyrochlores from the Lueshe carbonatite complex (Democratic Republic of Congo); a geochemical record of different alteration stages. J Afr Earth Sci 18:237–251
Nkurikiye L (1989) Le kaolin au Burundi: Contribution à sa caractérisation, sa genèse et son utilisation. Unpublished M. Sc. thesis, Burundi University
Notholt AJG (1999) Phosphate: a world monograph (a compilation of unfinished paper). British Geological Survey
Notholt AJG, Highley DE, Deans T (1990) Economic minerals in carbonatites and associated alkaline igneous rocks. Trans Inst Min Metall 99:B59–B80
Pell J (1996) Mineral deposits associated with carbonatites and related alkamine igneous rocks. In: Mitchell RH (ed) Undersaturated alkaline rocks: mineralogy, petrogenesis, and economic potential, vol 24, Short Course Series. Mineralogical Association of Canada, Winnipeg, pp 271–310
Penel G, Leroy G, Rey C, Sombret B, Huvenne JP, Bres E (1997) Infrared and Raman microspectrometry study of fluor-fluor-hydroxy and hydroxy-apatite powders. J Mater Sci 8:271–276
Penel G, Leroy G, Rey C, Bress E (1998) MicroRaman spectral study of the PO4 and CO3 vibrational modes in synthetic and biological apatites. Calcif Tissue Int 63:475–481
Pereira F, Bilal E (2012) Phosphoric acid extraction and rare earth recovery from apatites of the Brazilian phosphatic ores. Rom J Mineral Deposits 85:49–52
Songore T (1991) The Matongo phosphate deposits in Burundi. Fertil Res 30:151–153
Stormer JC, Pierson ML, Tacker RC (1993) Variation of F and Cl X-ray intensity due to anisotropic diffusion in apatite. Am Mineral 78:641–648
Sun SS (1982) Chemical composition and origin of the Earth’s primitive mantle. Geochim Cosmochim Acta 46:179–192
Swart PK, Burns SJ, Leder JJ (1991) Fractionation of the stable isotopes of oxygen and carbon in carbon dioxide during the reaction of calcite with phosphoric acid as a function of temperature and technique. Chem Geol 86:89–96
Tack L, De Paepe P, Deutsch S, Liégeois JP (1984) The alkaline plutonic complex of the Upper Ruvubu (Burundi): geology, age, isotopic geochemistry and implications for the regional geology of the Western rift. In: Klerkx J, Michot J (eds) African geology, Tervuren, pp 91–114
Tack L, Liégeois JP, André L, Navez J (1995) The Upper Ruvubu alkaline plutonic complex (Burundi). Annual Report of the Royal Museum for Central Africa, Tervuren, p 9
Tsuda H, Arends J (1994) Orientational micro-Raman spectroscopy on hydroxyapatite single crystals and human enamel crystallites. J Dent Res 73:1703–1710
Van der Beek P, Mbede E, Andriessen P, Delvaux D (1998) Denudation history of the Malawi and Rukwa Rift flanks (East African Rift System) from apatite fission track thermochronology. J Afr Earth Sci 26(3):363–385
Van Straaten (2002) Rocks for crops. Agrominerals of sub-Saharan Africa. International Centre for Research in Agroforestry, Nairobi, p 338
Vartiainen H, Paarma H (1979) Geological characteristics of the Sokli Carbonatite Complex, Finland. Econ Geol 74:1296–1306
Verwoed WJ (1986) Mineral deposits associated with carbonatites and alkaline rocks. In: Anhaeusser C, Maske S (eds) Mineral deposits of Southern Africa. Geol Soc S Afr, pp 2173–2191
Walter AV, Nahon D, Flicoteaux R, Girard JP, Melfi A (1995) Behaviour of major and trace elements and fractionation of REE under tropical weathering of a typical apatite-rich carbonatite from Brazil 136:591–602
Waychunas GA (2002) Apatite luminescence. In: Kohn MJ, Rakovan J, Hughes JM (eds) Reviews in mineralogy and geochemistry, vol 48, Phosphates. Mineralogical Society of America, Washington, pp 701–742
Wood SA (1990) The aqueous geochemistry of the rare-earth elements and yttrium: 1. Review of available low-temperature data for inorganic complexes and the inorganic REE speciation of natural waters. Chem Geol 82:159–186
Woolley AR (2001) Alkaline rocks and carbonatites of the world. Part 3: Africa. Geological Society of London, 372 p
Zaitsev V, Kogarko L (2012) Sources and perspectives of REE in the Lovozero massif (Kola Peninsula, Russia). European Mineralogical Conference. Abstract book, vol. 1, EMC2012-290, http://meetingorganizer.copernicus.org/EMC2012/EMC2012-290.pdf
Zheng YF (1999) Oxygen isotope fractionation in carbonate and sulfate minerals. Geochem J 33:109–126
Acknowledgments
The authors would like to warmly thank the MRAC for providing the study samples. Laurence Monin (MRAC) is thanked for her role in providing whole-rock analyses. Rénovat Nyandwi is thanked for his help and the preparation of the polished section. Chris Harris and an anonymous reviewer are sincerely thanked for their help to improve the quality of the manuscript. The authors are also grateful to Hartwig Frimmel and Bernd Lehmann for the editorial handling of this paper. Thomas Goovaerts is thanked for the English review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial handling: Lawrence P. Rozas
Electronic supplementary material
Below is the link to the electronic supplementary material.
Electronic supplementary material 1
Representative cathodoluminescence spectra of minerals in the Matongo phosphatic ore (operating conditions: 15 kV–500 μA, defocussed beam of approx. 4 mm diameter, spectra not corrected for system response). Upper spectra: Mn2+-activated calcite and Fe3+-activated albite and K-feldspar. The observed luminescence color is orange, red and pink, respectively. Lower spectra: Mn2+- and REE-activated primary fluorapatite (dominant blue-CL color) and Mn2+-activated secondary fluorapatite (green CL). The spectrum for the primary fluorapatite is representative for an average of its emission (blue-green and red-CL core and blue-violet rim). (JPEG 639 kb)
Electronic supplementary material 2
Raman spectra obtained on magmatic fluorapatites (a) and hydrothermal-supergene fluorapatites (b) from the Matongo phosphate deposit. Acquired using λ = 532 nn, power: 2 mW, acquisition time: 5 × 30 s, spectral range: 0–4500 cm−1. Peaks that are not directly related to the fluorapatite structure are present on the Raman spectra at the following frequencies (presented by decreasing intensities): ~2080 cm−1, ~3290 cm−1, in the 2500–2700 cm−1 range, in the 1750–1820 cm−1, and in the 1480–1530 cm−1 range. When the Raman shift is converted to wavelength (in nm), these unresolved peaks correspond fairly well to the REE emission lines. The most important are those of Sm3+ (at ~599, 644 and 652 nm), others would correspond to Eu3+ (multiple peaks in the 614–618 nm range), Tb3+ (one or two peaks at 587–588 nm), and Dy3+ (multiple peaks in the 577–579 m range). These wavelengths, which are typical of the fluorescence induced by the REE, can be related to the presence of these elements in the studied fluorapatite, what has been confirmed by in situ analyses on magmatic fluorapatites (Table 1) (PDF 2611 kb)
Table 1
Representative microprobe analyses (oxides in wt%) of apatite and monazite from the Matongo phosphate ore (XLSX 16 kb)
Table 2
Major element contents (in wt%), REE and trace elements (in ppm) of samples from the Matongo phosphate ore (XLSX 18 kb)
Rights and permissions
About this article
Cite this article
Decrée, S., Boulvais, P., Tack, L. et al. Fluorapatite in carbonatite-related phosphate deposits: the case of the Matongo carbonatite (Burundi). Miner Deposita 51, 453–466 (2016). https://doi.org/10.1007/s00126-015-0620-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00126-015-0620-1