Abstract
Purpose
To compare efficacy and patient outcome of wound infiltration with ropivacaine, lornoxicam, or their combination for control of pain following thyroid surgery.
Methods
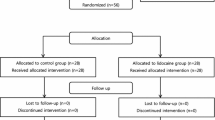
Eighty patients underwent thyroid surgery were randomly assigned to one of four groups. Before skin closure, local tissues were infiltrated with 12 mL saline in Group S, with 10 mL of ropivacaine 0.75% plus 2 mL saline in Group R, with 2 mL of lornoxicam (8 mg) plus 10 mL saline in Group L, and with 10 mL ropivacaine 0.75% plus 2 mL lornoxicam (8 mg) in Group RL. Pain scores, total and incremental meperidine consumption were recorded at 30 min, one, two, three, four, six, eight, 12, 18, and 24 hr postoperatively. Time to first analgesic requirement, patient satisfaction, and duration of hospital stay were also compared after surgery.
Results
The pain scores in Group RL were significantly lower in the first 12 hr than in Group S, and in the first four hours than in Groups R and L (P < 0.01). The time to first analgesic requirement was significantly longer (14.8 ± 8.4 hr vs 5.9 ± 5.2 hr;P < 0.01), the total pethidine consumption was significantly less than Group S (34.0 ± 33.0 mg vs 78.0 ± 29.8 mg; P<0.001), return of gastrointestinal function, ambulation time, length of hospital stay (P < 0.05) were significantly shorter, and patient satisfaction (P < 0.01) was significantly better in Group RL than in Group S (P < 0.05).
Conclusion
Wound infiltration with ropivacaine 0.75% plus lornoxicam 8 mg combination improved postoperative pain control and patient comfort, and decreased the need for opioids than the use of either drug alone.
Abstract
Objectif
Comparer ľefficacité ďune infiltration avec de la ropivacaïne, du lornoxicam ou leur combinaison après une opération de la thyroïde et comparer ľévolution des patients.
Méthode
Des patients devant être opérés à la thyroïde (80) ont été répartis en quatre groupes. Avant la fermeture cutanée, les tissus locaux ont été infiltrés avec 12 mL de solution saline dans le groupe S, 10 mL de ropivacaïne à 0,75 % plus 2 mL de solution saline dans le groupe R, 2 mL de lornoxicam (8 mg) plus 10 mL de solution saline dans le groupe L et 10 mL de ropivacaïne à 0,75 % plus 2 mL de lornoxicam (8 mg) dans le groupe RL. Les scores de douleur et la consommation totale et incrémentielle de mépéridine ont été notés à 30 min, puis à une, deux, trois, quatre, six huit, 12, 18 et 24 h après ľopération. Le moment de la première demande ďanalgésique, la satisfaction du patient et la longueur du séjour ont été comparés.
Résultats
Les scores de douleurs ont été significativement plus bas chez les patients du groupe RL que chez ceux du groupe S pendant les 12 premières heures et que chez ceux des groupes R et L pendant les quatre premières heures (P < 0,01). Le moment de la première demande ďanalgésique a été plus tardif (14,8 ± 8,4 h vs 5,9 ± 5,2 h ; P < 0,01), la consommation totale de péthidine a été plus basse que dans le groupe S (34,0 ± 33,0 mg vs 78,0 ± 29,8 mg ; P < 0,001), le retour de la fonction gastro-intestinale et de la marche a été plus précoce, la longueur du séjour hospitalier (P < 0,05) plus courte et la satisfaction des patients (P < 0,01) meilleure dans le groupe RL que dans le groupe S (P < 0,05).
Conclusion
Ľinfiltration dans le site ďincision avec une combinaison de ropivacaïne à 0,75 % et de 8 mg de lornoxicam a amélioré le contrôle postopératoire de la douleur et le confort des patients et a diminué les besoins ďopioïdes par rapport à ľusage ďun seul médicament.
Article PDF
Similar content being viewed by others
References
Daou R. Thyroidectomy without drainage. Chirurgie 1997; 122: 408–10.
Dieudonne N, Gomola A, Bonnichon P, Ozier YM. Prevention of postoperative pain after thyroid surgery: a double-blind randomized study of bilateral superficial cervical plexus blocks. Anesth Analg 2001; 92: 1538–42.
Radhofer-Welte S, Rabasseda X. Lornoxicam, a new potent NSAID with an improved tolerability profile. Drugs Today 2000; 36: 55–76.
Balfour JA, Fitton A, Barradell LB. Lornoxicam. A review of its pharmacology and therapeutic potential in the management of painful and inflammatory conditions. Drugs 1996; 51: 639–57.
Rosenow DE, van Krieken F, Stolke D, Kursten FW. Intravenous administration of lornoxicam, a new NSAID, and pethidine for postoperative pain. A placebo-controlled pilot study. Clin Drug Invest 1996; 11: 11–9.
Bianconi M, Ferraro L, Ricci R, et al. The pharmacokinetics and efficacy of ropivacaine continuous wound instillation after spine fusion surgery. Anesth Analg 2004; 98: 166–72.
Horn EP, Schroeder F, Wilhelm S, et al. Wound infiltration and drain lavage with ropivacaine after major shoulder surgery. Anesth Analg 1999; 89: 1461–6.
Hannibal K, Galatius H, Hansen A, Obel E, Ejlersen E. Preoperative wound infiltration with bupivacaine reduces early and late opioid requirement after hysterectomy. Anesth Analg 1996; 83: 376–81.
Dierking GW, Ostergaard E, Ostergard HT, Dahl JB. The effects of wound infiltration with bupivacaine versus saline on postoperative pain and opioid requirements after herniorrhaphy. Acta Anaesthesiol Scand 1994; 38: 289–92.
Sztark F, Malgat M, Dabadie P, Mazat JP. Comparison of the effects of bupivacaine and ropivacaine on heart cell mitochondrial bioenergetics. Anesthesiology 1998; 88: 1340–9.
Scott DB, Lee A, Fagan D, Bowler GM, Bloomfield P, Lundh R. Acute toxicity of ropivacaine compared with that of bupivacaine. Anesth Analg 1989; 69: 563–9.
Kopacz DJ, Carpenter RL, Mackey DC. Effect of ropivacaine on cutaneous capillary blood flow in pigs. Anesthesiology 1989; 71: 69–74.
Martinsson T, Oda T, Fernvik E, Roempke K, Dalsgaard CJ, Svensjo E. Ropivacaine inhibits leukocyte rolling, adhesion and CD11b/CD18 expression. J Pharmacol Exp Ther 1997; 283: 59–65.
Visalyaputra S, Sanansilp V, Pechpaisit N, et al. Postoperative analgesic effects of intravenous lornoxicam and morphine with pre-emptive ropivacaine skin infiltration and preperitoneal instillation after transabdominal hysterectomy. J Med Assoc Thai 2002; 85(Suppl 3): S1010–6.
Georgiadou TH, Sfyra E, Georgiou M, Foulidou A, Papaioannou V, Kanakoudis F. Postoperative wound infiltration with chirocaine plus lornoxicam for pain relief after cholecystectomy. Eur J Anaesthesiol 2003; 20(Suppl 30): A-689 (abstract).
Jorgensen H, Fomsgaard JS, Dirks J, Wetterslev J, Andreasson B, Dahl JB. Effect of peri- and postoperative epidural anaesthesia on pain and gastrointestinal function after abdominal hysterectomy. Br J Anaesth 2001; 87: 577–83.
Scott RM, Jennings PN. Rectal diclofenac analgesia after abdominal hysterectomy. Aust NZ J Obstet Gynaecol 1997; 37: 112–4.
Kam PC, Power I. New selective COX-2 inhibitors. Pain Reviews 2000; 7: 3–13.
Fishman S, Borsook D. Opioids in pain management.In: Benzon H, Raja S, Molloy RE, Strichartz G (Eds). Essentials of Pain Medicine and Regional Anesthesia. New York: Churchill Livingstone; 1999: 51–4.
Ilias W, Jansen M. Pain control after hysterectomy: an observer-blind, randomised trial of lornoxicam versus tramadol. Br J Clin Pract 1996; 50: 197–202.
Trampitsch E, Pipam W, Moertl M, et al. Preemptive randomized, double-blind study with lornoxicam in gynecological surgery (German). Schmerz 2003; 17: 4–10.
Bianconi M, Ferraro L, Traina GC, et al. Pharmacokinetics and efficacy of ropivacaine continuous wound instillation after joint replacement surgery. Br J Anaesth 2003; 91: 830–5.
Gottschalk A, Burmeister MA, Radtke P, et al. Continuous wound infiltration with ropivacaine reduces pain and analgesic requirement after shoulder surgery. Anesth Analg 2003; 97: 1086–91.
Klein JR, Heaton JP, Thompson JP, Cotton BR, Davidson AC, Smith G. Infiltration of the abdominal wall with local anaesthetic after total abdominal hysterectomy has no opioid-sparing effect. Br J Anaesth 2000; 84: 248–9.
Cobby TF, Reid MF. Wound infiltration with local anaesthetic after abdominal hysterectomy. Br J Anaesth 1997; 78: 431–2.
Gozal Y, Shapira SC, Gozal D, Magora F. Bupivacaine wound infiltration in thyroid surgery reduces postoperative pain and opioid demand. Acta Anaesthesiol Scand 1994; 38: 813–5.
Lacoste L, Thomas D, Kraimps JL, et al. Postthyroidectomy analgesia: morphine, buprenorphine, or bupivacaine? J Clin Anesth 1997; 9: 189–93.
Staunstrup H, Ovesen J, Larsen UT, Elbaek K, Larsen U, Kroner K. Efficacy and tolerability of lornoxicam versus tramadol in postoperative pain. J Clin Pharmacol 1999; 39: 1–8.
Rosenow DE, Albrechtsen M, Stolke D. A comparison of patient-controlled analgesia with lornoxicam versus morphine in patients undergoing lumbar disk surgery. Anesth Analg 1998; 86: 1045–50.
Watcha MF, White PF. Postoperative nausea and vomiting. Its etiology, treatment, and prevention. Anesthesiology 1992; 77: 162–84.
Wang JJ, Ho ST, Lee SC, Liu YC, Ho CM. The use of dexamethasone for preventing postoperative nausea and vomiting in females undergoing thyroidectomy: a dose-ranging study. Anesth Analg 2000; 91: 1404–7.
Wang JJ, Ho ST, Lee SC, Liu YC, Liu YH, Liao YC. The prophylactic effect of dexamethasone on postoperative nausea and vomiting in women undergoing thyroidectomy: a comparison of droperidol with saline. Anesth Analg 1999; 89: 200–3.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karamanlioglu, B., Turan, A., Memis, D. et al. Infiltration with ropivacaine plus lornoxicam reduces postoperative pain and opioid consumption. Can J Anesth 52, 1047–1053 (2005). https://doi.org/10.1007/BF03021603
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03021603