Summary
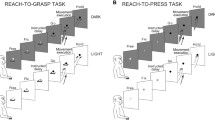
Neuronal activity was studied in the primary (M1), supplementary (M2), dorsal premotor (PMd), and ventral premotor (PMv) cortex of awake, behaving rhesus monkeys. The animals performed forelimb movements to three targets, each approached by three different types of trajectories. With one trajectory type, the monkey moved its hand straight to the target, with another, the path curved in a clockwise direction, and with a third, the path curved in a counter-clockwise direction. We examined whether neuronal activity in these areas exclusively reflects a hand movement's net distance and direction or, alternatively, whether other factors also influence cortical activity. It was found that neuronal activity during all phases of a trial reflects aspects of movement in addition to target location. Among these aspects may be selection of an integrated motor act from memory, perhaps specifying the entirety of a path by which the hand moves to a target.
Similar content being viewed by others
References
Abend W, Bizzi E, Morasso P (1982) Human arm trajectory formation. Brain 105: 331–348
Alexander GE, Crutcher MD (1990) Neural representation of the target (goal) of visually guided arm movements in three motor areas of the monkey. J Neurophysiol 64: 164–178
Amos TM, Vermeersch CH, Park S-K, Ebner, TJ (1988). Principle [sic] component analysis of the responses of premotor cortical neurons to movement in two dimensions. Soc Neurosci Abstr 14: 343
Bucy PC, Fulton JF (1933) Ipsilateral representation in the motor and premotor cortex of monkeys. Brain 56: 318–342
Caminiti R, Johnson PB, Pastore RA, Urbano A (1989) The cortical representation of a coordinate system for the planning and execution of arm movements in space. In: Jeannerod M, Joseph JP (eds) Cortical control of sensory motor integration. Abstr EBBS workshop European Brain and Behavior Society, Lyon, France, p 25
Caminiti R, Johnson PB, Urbano A (1990) Making arm movements within different parts of space: dynamic aspects in the primate motor cortex. J Neurosci 10: 2039–2058
Crammond DJ, Kalaska JF (1989a) Neuronal discharge during an instructed-delay period. II. Premotor area 6. In: Jeannerod M, Joseph JP (eds) Cortical control of sensory-motor integration. Abstr EBBS workshop European Brain and Behavior Society, Lyon, France, p 15
Crammond DJ, Kalaska JF (1989b) Neuronal activity in primate parietal cortex area 5 varies with intended movement direction during an instructed-delay period. Exp Brain Res 76: 458–462
Crammond DJ, Kalaska JF (1989c) Comparison of cell activity in cortical areas 6, 4 and 5 recorded in an instructed delay task. Soc Neurosci Abstr 15: 786
Crutcher MD, Alexander GE (1988) Supplementary motor area (SMA): coding of both preparatory and movement-related neural activity in spatial rather than joint coordinates. Soc Neurosci Abstr 14: 342
Evarts EV (1968) Relation of pyramidal tract activity to force exerted during voluntary movement. J Neurophysiol 31: 14–27
Evarts EV (1969) Activity of pyramidal tract neurons during postural fixation. J Neurophysiol 32: 375–385
Fulton JF (1935) Definition of the motor and premotor areas. Brain 58: 311–216
Gentilucci M, Scandolara C, Pigarev IN, Rizzolatti G (1983) Visual responses in the postarcuate cortex (area 6) of the monkey that are independent of eye position. Exp Brain Res 50: 464–468
Georgopoulos AP (1987) Cortical mechanisms subserving reaching. In: Bock G, O'Connor M, Marsh J (eds) Motor areas of the cerebral cortex. Ciba Foundation symposium 132. John Wiley & Sons, Chichester, pp 125–141
Georgopoulos AP, Kalaska JF, Caminiti R, Massey JT (1982) On the relations between the direction of two-dimensional arm movements and cell discharge in primate motor cortex. J Neurosci 11: 1527–1537
Georgopoulos AP, Caminiti R, Kalaska JF, Massey JT (1983) Spatial coding of movement: a hypothesis concerning the coding of movement direction by motor cortical populations. Exp Brain Res Suppl 7: 327–336
Georgopoulos AP, Kalaska JF, Caminiti R (1985) The relations between two dimensional arm movement and single-cell discharge in motor cortex and area 5: movement direction vs. movement end-point. Exp Brain Res Suppl 10: 175–183
Georgopoulos AP, Kettner RE, Schwartz B (1988) Primate motor cortex and.free arm movements to visual targets in threedimensional space. II. Coding of the direction of movement by a neuronal population. J Neurosci 8: 2928–2937
Georgopoulos AP, Crutcher MD, Schwartz AB (1989) Cognitive spatial-motor processes. 3. Motor cortical prediction of movement direction during an instructed delay period. Exp Brain Res 75: 183–194
Godschalk M, Lemon RN, Kuypers HGJM, van der Steen J (1985) The involvement of monkey premotor cortex neurones in preparation of visually cued arm movements. Behav Brain Res 18: 143–157
Hocherman S, Wise SP (1989) Movement trajectory or goal representation in primate premotor and primary motor cortex. Soc Neurosci Abstr 15: 789
Hocherman S, Wise SP (1990) Trajectory-selective neuronal activity in the motor cortex of rhesus monkeys (Macaca mulatto). Behav Neurosci 104: 495–499
Humphrey DR (1986) Representation of movements and muscles within the primate precentral motor cortex: historical and current perspective. Fed Proc 45: 2687–2699
Humphrey DR, Schmidt EM, Thompson WD (1970) Predicting measures of motor performance from multiple cortical spike trains. Science 179: 758–762
Kalaska JF, Cohen DAD, Hyde ML, Prud'homme M (1989) A comparison of movement direction-related versus load directionrelated activity in primate motor cortex, using a two-dimensional reaching task. J Neurosci 9: 2080–2102
Karluk D, Ebner TJ (1989) Spatial representation of movement distance and direction in the premotor cortex. Soc Neurosci Abstr 15: 787
Kurata K (1988) Motor programming in premotor cortex of the monkeys. Abstr Twelfth International Symposium of the Taniguchi Foundation. Toyobo Co, Katata, Ohtsu, Japan, pp 13–14
Kurata K, Wise SP (1988) Premotor cortex of rhesus monkeys: setrelated activity during two conditional motor tasks. Exp Brain Res 69: 327–343
Kwan HC, MacKay WA, Murphy JT, Wong YC (1981) Distribution of responses to visual cues for movement in precentral cortex of awake primates. Neurosci Lett 24: 123–128
Kwan HC, MacKay WA, Murphy JT, Wong YC (1985) Properties of visual cue responses in primate precentral cortex. Brain Res 343: 24–35
Mitz AR, Wise SP, Godschalk M (1989) Learning-dependent activity in premotor cortex of rhesus monkeys. Soc Neurosci Abstr 15: 787
Moll L, Kuypers HGJM (1977) Premotor cortical ablations in monkeys: contralateral changes in visually guided reaching behavior. Science 198: 317–319
Murray GM and Sessle BJ (1989) Coding of tongue movement direction by neurones in tongue region of motor cortex (MI) of awake monkeys (M.fascicularis). Soc Neurosci Abstr 15: 788
Mussa-Ivaldi FA (1988) Do neurons in the motor cortex encode movement direction? An alternative hypothesis. Neurosic Lett 91: 106–111
Passingham RE (1987) Two cortical systems for directing movement. In: Bock G, O'Connor M, Marsh J (eds) Motor areas of the cerebral cortex. Ciba Foundation Symposium 132. John Wiley & Sons, Chichester, pp 151–164
Passingham RE (1988) Premotor cortex and preparation for movement. Exp Brain Res 70: 590–596
Rea GL, Ebner TJ, Bloedel JR (1987). Evaluations of combined premotor and supplementary motor cortex lesions on a visually guided arm movement. Brain Res 418: 58–67
Riehle A, Requin J (1989) Monkey primary motor and premotor cortex: single-cell activity related to prior information about direction and extent of an intended movement. J Neurophysiol 61: 534–549
Rizzolatti G, Gentilucci M, Fogassi L, Luppino G, Matelli M, Ponzoni-Maggi S (1987) Neurons related to goal-directed motor acts in inferior area 6 of the macaque monkey. Exp Brain Res 67: 220–224
Rizzolatti G, Camarda R, Fogassi L, Gentilucci M, Luppino G, Matelli M (1988) Functional organization of inferior area 6 in the macaque monkey. II. Area F5 and the control of distal movements. Exp Brain Res 71: 491–507
Schwartz AB, Anderson BJ (1989) Motor cortical images of sinusoidal trajectories. Soc Neurosci Abstr 15: 788
Schwartz AB, Kettner RE, Georgopoulos AP (1988) Primate motor cortex and free arm movements to visual targets in three-dimensional space. I. Relations between single cell discharge and direction of movement. J Neurosci 8: 2913–2927
Tanji J, Evarts EV (1976) Anticipatory activity of motor cortex neurons in relation to direction of an intended movement. J Neurophysiol 39: 1062–1068
Thach WT (1978) Correlation of neural discharge with pattern and force of muscular activity, joint position, and direction of intended next movement in motor cortex and cerebellum. J Neurophysiol 41: 654–676
Traverse J, Latto R (1986) Impairments in route negotiation through a maze after dorsolateral frontal, inferior parietal or premotor lesions in cynomolgus monkeys. Behav Brain Res 20: 203–215
Wannier TMJ, Maier MA, Hepp-Reymond M-C (1989) Responses of motor cortex neurons to visual stimulation in the alert monkey. Neurosic Lett 98: 63–68
Weinrich M, Wise SP (1982) The premotor cortex of the monkey. J Neurosci 2: 1329–1345
Weinrich M, Wise SP, Mauritz K-H (1984) A neurophysiological analysis of the premotor cortex of the monkey. Brain 107: 385–414
Wise SP (1984) Nonprimary motor cortex and its role in the cerebral control of movement. In: Edelman G, Cowan WM, Gall E (eds) Dynamic aspects of neocortical function. Wiley, New York, pp 525–555
Wise SP (1985) The primate premotor cortex: past, present, and preparatory. Ann Rev Neurosci 8: 1–19
Wise SP, Mauritz K-H (1985) Set-related neuronal activity in the premotor cortex of rhesus monkeys: effects of changes in motor set. Proc R Soc Lond B 223: 331–354
Woolsey CN, Settlage PH, Meyer DR, Sencer W, Pinto Hamuy T, Travis AM (1952) Patterns of localization in precentral and “supplementary” motor areas and their relation to the concept of a premotor area. Res Publ Assoc Res Nerv Ment Dis 30: 238–264
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hocherman, S., Wise, S.P. Effects of hand movement path on motor cortical activity in awake, behaving rhesus monkeys. Exp Brain Res 83, 285–302 (1991). https://doi.org/10.1007/BF00231153
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00231153